Ch1 : Metallurgy
MOST REPEATED QUESTIONS
Number next to question tells how many times the question has been repeated in the past few years
📌 TWO MARKS
1.Explain Mond’s process of refining Nickel?(7)
- The impure nickel is heated in a stream of carbon monoxide at around 350 K.
- The nickel reacts with the CO to form a highly volatile nickel tetracarbonyl.
- The solid impurities are left behind.

- On heating the nickel tetracarbonyl around 460 K, the complex decomposes to give pure metal.

Draw above equation in reverse order
2.What are the differences between minerals and ores?(4)
| Minerals | Ores |
| Naturally occurring substances obtained by mining which contain the metals in free state or in the form of compounds like oxides, sulphides etc. are called minerals. | Minerals that contain high percentage of metal from which it can be extracted conveniently and economically are called ores. |
| All minerals are not ores | All ores are minerals |
| Ex : Clay – Mineral of aluminium | Ex : Bauxite – Ore of aluminium |
3.Describe the role of the following in the process mentioned. (4)
(i) Silica in the extraction of copper.
Silica acts as an acidic flux to remove FeO as slag FeSiO3. in the extraction of copper
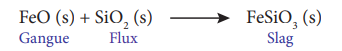
(ii) Cryolite in the extraction of aluminium:
Cryolite serves as an added impurity and lowers the melting point of the Al2O3 .
iii) Iodine in the refining of Zirconium:
First Iodine forms a Volatile tetraiodide with impure metal, which decomposes to give pure metal. Impure zirconium metal is heated in an evacuated vessel with iodine to form the volatile zirconium tetraiodide (ZrI4). The impurities are left behind, as they do not react with iodine.
iv) Sodium cyanide in froth floatation:
Sodium cyanide acts as a depressing agent in froth floatation process. When a sulphide ore of a metal of interest contains other metal sulphides the depressing agent sodium cyanide selectively prevent other metal sulphides from coming to the froth.
4.What is the role of Limestone in the extraction of Iron from its oxide Fe2O3?(3)
In this extraction, a basic flux, limestone (CaO) is used. Since the silica gangue present in the ore is acidic in nature, the limestone combines with it to form calcium silicate (slag).
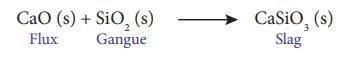
5.What are the various steps involved in extraction of pure metals from their ores ? (b/b : 2) (2)
- Coversion of the ore into oxides of the metal of Interest
- Reduction of the metal oxides to elemental Metals
6.Which type of ore can be separated from froth floatation method ?Give two examples.(2)
Sulphide ores can be concentrated by froth floatation method.
E.x : Galena (PbS), Zinc blende (ZnS)
7.Write short notes on Auto reduction?pg10(2)
Simple roasting of some of the ores give the crude metal. In such cases, the use of reducing agents is not necessary. For example, mercury is obtained by roasting of its ore cinnabar (HgS)

8.Define Gangue and slag?(2)
(i) Gangue
- The non metallic impurities, rocky materials and siliceous matter present in the ores are called gangue.
- E.x : SiO2 is the gangue present in the iron ore Fe2O3.
ii) Slag
Slag is a fusible chemical substance formed by the reaction of gangue with a flux.

📌 THREE MARKS
1.Explain zone refining process with an example?(4)
- This method is based on the principles of fractional crystallisation.
- When an impure metal is melted and allowed to solidify, the impurities will prefer to be in the molten region.
- In this process the impure metal is taken in the form of a rod.
- One end of the rod is heated using a mobile induction heater which results in melting of the metal on that portion of the rod.
- When the heater is slowly moved to the other end the pure metal crystallises while the impurities will move on to the adjacent molten zone formed due to the movement of the heater.
- As the heater moves further away, the molten zone containing impurities also moves along with it.
- The process is repeated several times by moving the heater in the same direction again and again to achieve the desired purity level.
- This process is carried out in an inert gas atmosphere to prevent the oxidation of metals .
- Elements such as germanium (Ge), silicon (Si) and galium (Ga) that are used as semiconductor are refined using this process.
2.List out the observations from Ellingham diagram?(2)
- For most of the metal oxide formation, the slope is positive.
- Oxygen gas is consumed during the formation of metal oxides which results in the decrease in randomness. Hence, ΔS becomes negative and it makes the term, TΔS positive in the straight line equation.
ΔG = ΔH – TΔS
- The graph for the formation of carbon monoxide is a straight line with negative slope. In this case ΔS is positive as 2 moles of CO gas is formed by the consumption of one mole of oxygen gas. It indicates that CO is more stable at higher temperature.
- As the temperature increases, generally ΔG value for the formation of the metal oxide become less negative and becomes zero at a particular temperature. Below this temperature, ΔG is negative and the oxide is stable and above this temperature ΔG is positive. This general trend suggests that metal oxides become less stable at higher temperature and their decomposition becomes easier.
- There is a sudden change in the slope at a particular temperature for some metal oxides like MgO, HgO. This is due to the phase transition (melting or evaporation).
3.Give the limitations of ellingham diagram?(2)
1. Ellingham diagram is constructed based only on thermodynamic considerations. It gives information about the thermodynamic feasibility of a reaction. It does not tell anything about the rate of the reaction. More over, it does not give any idea about the possibility of other reactions that might be taking place.
2. The interpretation of ΔG is based on the assumption that the reactants are in equilibrium with the products which is not always true.
📌 FIVE MARKS
1.Explain the principle of electrolytic refining with an example?(5)
The crude metal is refined by electrolysis, It is carried out in an electrolytic cell.
Anode : The rods of impure metal
Cathode : Thin strips of pure metal are used as cathode
Electrolyte : Aqueous solution of the salts of the metal.
The metal of interest dissolves from the anode, pass into the solution while the same amount of metal ions from the solution will be deposited at the cathode. During electrolysis, the less electropositive impurities in the anode, settle down at the bottom and are removed as anode mud.
Electrolytic refining of silver
- Cathode : Pure silver
- Anode : Impure silver rods
- Electrolyte : Acidified aqueous solution of silver nitrate.
When a current is passed through the electrodes the following reactions will take place
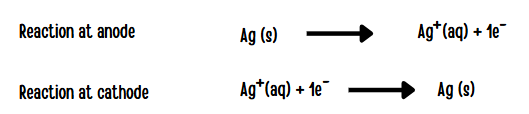
During electrolysis, at the anode the silver atoms lose electrons and enter the solution. The positively charged silver cations migrate towards the cathode and get discharged by gaining electrons and deposited on the cathode.
Other metals such as copper, zinc etc.,can also be refined by this process in a similar manner
2.Explain the method of froth floatation(4)
- This method is commonly used to concentrate sulphide ores such as galena (PbS), zinc blende (ZnS) etc
- The metallic ore particles which are wetted by oil can be separated from gangue.
- In this method, the crushed ore is suspended in water and mixed with frothing agent such as pine oil, eucalyptus oil etc.
- A small quantity of sodium ethyl xanthate which acts as a collector is also added.
- A froth is generated by blowing air through this mixture.
- The collector molecules attach to the ore particle and make them water repellent.
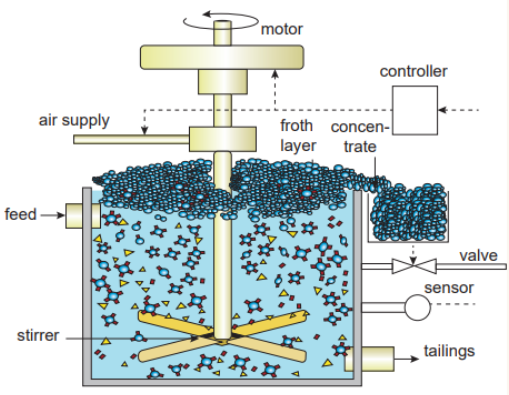
As a result, ore particles, wetted by the oil, rise to the surface along with the froth. The froth is skimmed off and dried to recover the concentrated ore. The gangue particles that are preferentially wetted by water settle at the bottom.
3.Explain gravity separation?(3)
- In this method, the ore having high specific gravity is separated from the gangue that has low specific gravity by simply washing with running water.
- Ore is crushed to a finely powdered form and treated with rapidly flowing current of water.
- During this process the lighter gangue particles are washed away by the running water.
- This method is generally applied to concentrate the native ore such as gold and oxide ores such as haematite (Fe O ), tin stone (SnO ) etc.
4.Explain the electro metallurgy of alluminium?or Hall-Heroult method?(3)
Cathode : Iron tank lined with carbon.
Anode : The carbon blocks.
Electrolyte: A 20% solution of alumina, obtained from the bauxite ore is mixed with molten cryolite.
- About 10% calcium chloride is also added to the solution. Here calcium chloride helps to lower the melting point of the mixture
Temperature : Above 1270 K


