p-Block Elements-I
MOST REPEATED QUESTIONS
Number next to question tells how many times the question has been repeated in the past few years
📌 TWO MARKS
1.What are the uses of silicones?(6)
- Silicones are used for low temperature lubrication and in vacuum pumps, high temperature oil baths etc…
- They are used for making water proofing clothes
- They are used as insulting material in electrical motor and other appliances
- They are mixed with paints and enamels to make them resistant towards high temperature, sunlight, dampness and chemicals.
2.Write a note on fischer tropsch synthesis?(5)
The reaction of carbon monoxide with hydrogen at a pressure of less than 50 atm using metal catalysts at 500 – 700 K yields saturated and unsaturated hydrocarbons.

3.How will you identify borate radical? Or Ethyl Borate Test ?(4)
When boric acid or borate salt is heated with ethyl alcohol in presence of conc. sulphuric acid, an ester, triethylborate is formed. The vapour of this ester burns with a green edged flame and this reaction is used to identify the presence of borate.
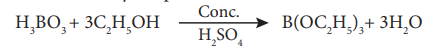
4.Write the uses of Borax?(4)
- Borax is used for the identification of coloured metal ions
- In the manufacture of optical and borosilicate glass, enamels and glazes for pottery
- It is also used as a flux in metallurgy and also acts as a preservative
5.What is inert pair effect?Pg52(4)
In heavier post transition metals.the outer s electron(ns) have a tendency to remain inert and show reluctance to take part in the bonding.which is known as inert pair effect
📌 THREE MARKS
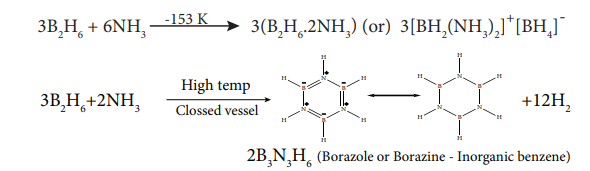
1.Write the preparation of Borazole/inorganic benzene?(4)
When diborane treated with excess ammonia at low temperatures diborane gives diboranediammonate. On heating at higher temperatures it gives borazole.
2.What is catenation? describe briefly the catenation property of carbon?Ln2(4)
Catenation is an ability of an element to form chain of atoms.
Catenation property of carbon.
- The valency of element is greater than or equal to two,
- Element should have an ability to bond with itself
- The self bond must be as strong as its bond with other elements
- kinetic inertness of catenated compound towards other molecules.
Carbon possesses all the above properties and forms a wide range of compounds with itself and with other elements such as H, O, N, S and halogen
3.Write a short note on anomalous properties of the first element of p-Block?Ln2(2)
The following factors are responsible for this anomalous behaviour.
- Small size of the first member
- High ionisation enthalpy and high electronegativity
- Absence of d orbitals in their valance shell
4.Explain Mcafee process?(2)
Aluminium chloride is obtained by heating a mixture of alumina and coke in a current of chlorine.

On industrial scale it is prepared by chlorinating aluminium around 1000 K

📌 FIVE MARKS
1.Describe the structure of diborane? (5)
- In diborane two BH2 units are linked by two bridged hydrogens.
- Therefore, it has eight B-H bonds.
- The four terminal B-H bonds are normal covalent bonds (two centre – two electron bond or 2c-2e bond).
- The remaining four electrons have to be used for the bridged bonds. i.e. two three centred B-H-B bonds utilise two electrons each.
- Hence, these bonds are three centre- two electron bonds (3c-2e).
- The bridging hydrogen atoms are in a plane.
- In diborane, the boron is sp3 hybridised.
- Three of the four sp3 hybridised orbitals contains single electron and the fourth orbital is empty.
- Two of the half filled hybridised orbitals of each boron overlap with the 1s orbitals of two hydrogens to form four terminal 2c-2e bonds, leaving one empty and one half filled hybridised orbitals on each boron.
- The Three centre – two electron bonds, B-H-B bond formation involves overlapping the half filled hybridised orbital of one boron, the empty hybridised orbital of the other boron and the half filled 1s orbital of hydrogen.
2.Write a note on zeolites? (4)
- Zeolites are three-dimensional crystalline solids containing aluminium, silicon, and oxygen in their regular three dimensional framework.
- They are hydrated sodium alumino silicates with general formula Na2O.(Al2O3 ).x(SiO2 ).yH2O (x=2 to 10; y=2 to 6).
- Zeolites have a three dimensional crystalline structure looks like a honeycomb consisting of a network of interconnected tunnels and cages.
- Water molecules moves freely in and out of these pores but the zeolite framework remains rigid.
- Another special aspect of this structure is that the pore/channel sizes are nearly uniform, allowing the crystal to act as a molecular sieve.
