12th Chemistry Chapter 5 Important 2, 3, 5 Mark Questions 2026
Coordination Chemistry
MOST REPEATED QUESTIONS
Number next to question tells how many times the question has been repeated in the past few years
📌 TWO MARKS
1.[Ti(h20)6]3+is coloured while [Sc(H20)6]3+is colourless.why?(6)
- Ti : 3d2 4s2
- Ti3+ : 3d1 4s0
- [Ti(h20)6]3+ has one unpaired electron so it is coloured.
- Sc : 3d14s2
- Sc3+ : 3d0
- Sc(H20)6]3+ has no unpaired electrons so it is colourless.
2.What is meant by the term “coordination Number”?what is the cordination number of atoms in a bcc structure?
The number of ligand donor atoms bonded to a central metal ion in a complex is called the coordination number of the metal. In other words, the coordination number is equal to the number of σ-bonds between ligands and the central atom.
coordination Number of atoms in bcc is 8
3.What are the limitations of VB theory?(2)
- It does not explain the colour of the complex.
- It considers only the spin magnetic moments and does not consider the other components of magnetic moments.
- It does not provide a quantitative explanation as to why certain complexes are inner orbital complexes and the others are outer orbital complexes for the same metal. For example, [Fe(CN)6]4- is diamagnetic (low spin) whereas [FeF6]4- is paramagnetic (high spin).
📌 THREE MARKS
1.In the complex [Pt(NH3)3(NO2)]CL identify the following?
- A)Centarl metal atom/ion : Pt(Platinum)
- B)Ligands : NH3 , NO2
- C)Coordination Number : 4
- D)IUPAC Name : triamminechloronitroplatinum(II) chloride.
- E)Coordination entity : Pt(NH3)3(NO2)
2.In the complex, [Pt(NO2)(H2O)(NH3)2]Br , identify the following,
- A)Centarl metal atom/ion : Pt
- B)Ligands : NO2– Negative ligand , H2O – Natural Ligand , NH3 – Neutral Ligand
- C)Coordnation Entity : Pt(NO2)(H2O)(NH3)2
- D)Oxidation Number of central metal ion :
Pt(NO2)(H2O)(NH3)2]Br
- X + (-1) +( 0) + (0) +( -1) = 0
- X = 1+1
- = 2
- E)Coordination Number : 4 (1- No2 , 1 – H20 , 2 – NH3 = 4 Ligands attached)
2.In an octahedral crystal,draw and explain the figure to show splitting of d-orbitals?
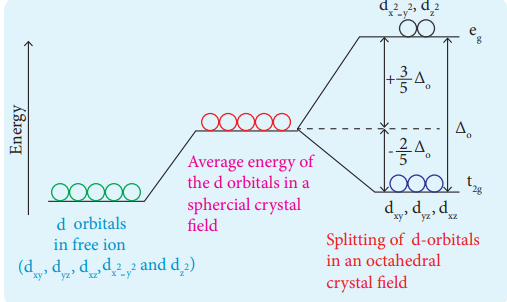
During crystal field splitting in octahedral field, in order to maintain the average energy of the orbitals (barycentre) constant, the energy of the orbitals dx2 – y 2 and dz2 (represented as eg orbitals) will increase by 3/5Δo while that of the other three orbitals dxy, dyz and dzx (represented as t2g orbitals) decrease by 2/5Δo . Here, Δo represents the crystal field splitting energy in the octahedral field.
4.Explain ionisation isomerism with an example?
- This type of isomers arises when an ionisable counter ion (simple ion) itself can act as a ligand.
- The exchange of such counter ions with one or more ligands in the coordination entity will result in ionisation isomers.
- These isomers will give different ions in solution.
- For example, consider the coordination compound [Pt(en)2 Cl2 ]Br2 .
- In this compound, both Br– and Cl– have the ability to act as a ligand and the exchange of these two ions result in a different isomer [Pt(en)2 Br2 ]Cl2 .
- In solution the first compound gives Br– ions while the later gives Cl –ions and hence these compounds are called ionisation isomers.
6.Write short note on – π-Back bonding in metal carbonyl?
- In metal carbonyls, the bond between metal atom and the carbonyl ligand consists of two components.
- The first component is an electron pair donation from the carbon atom of carbonyl ligand into a vacant d-orbital of central metal atom.
- This electron pair donation forms
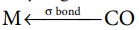
sigma bond.
- This sigma bond formation increases the electron density in metal d orbitals and makes the metal electron rich.
- In order to compensate for this increased electron density, a filled metal d-orbital interacts with the empty π* orbital on the carbonyl ligand and transfers the added electron density back to the ligand.
- This second component is called π-back bonding .
📌 FIVE MARKS
1.Explain werner’s theory of coordination compounds/postulates of werner theory?
- Most of the elements exhibit, two types of valence namely primary valence and secondary valence and each element tend to satisfy both the valences.In modern terminology, the primary valence is referred as the oxidation state of the metal atom and the secondary valence as the coordination number. For example, according to Werner, the primary and secondary valences of cobalt are 3 and 6 respectively.
- The primary valence of a metal ion is positive in most of the cases and zero in certain cases. They are always satisfied by negative ions. For example in the complex CoCl3 .6NH3 , The primary valence of Co is +3 and is satisfied by 3Cl- ions.
- The secondary valence is satisfied by negative ions, neutral molecules, positive ions or the combination of these. For example, in CoCl3 .6NH3 the secondary valence of cobalt is 6 and is satisfied by six neutral ammonia molecules, whereas in CoCl3 .5NH3 the secondary valence of cobalt is satisfied by five neutral ammonia molecules and a Cl– ion.
- According to Werner, there are two spheres of attraction around a metal atom/ion in a complex. The inner sphere is known as coordination sphere and the groups present in this sphere are firmly attached to the metal. The outer sphere is called ionisation sphere. The groups present in this sphere are loosely bound to the central metal ion and hence can be separated into ions upon dissolving the complex in a suitable solvent.
2.Main assumptions of VB Theory(Valency Bond Theory)?
- The ligand → metal bond in a coordination complex is covalent in nature. It is formed by sharing of electrons (provided by the ligands) between the central metal atom and the ligand.
- Each ligand should have at least one filled orbital containing a lone pair of electrons.
- In order to accommodate the electron pairs donated by the ligands, the central metal ion present in a complex provides required number (coordination number) of vacant orbitals.
- These vacant orbitals of central metal atom undergo hybridisation, the process of mixing of atomic orbitals of comparable energy to form equal number of new orbitals called hybridised orbitals with same energy.
- The vacant hybridised orbitals of the central metal ion, linearly overlap with filled orbitals of the ligands to form coordinate covalent sigma bonds between the metal and the ligand.
- The hybridised orbitals are directional and their orientation in space gives a definite geometry to the complex ion.
- In the octahedral complexes, if the (n-1) d orbitals are involved in hybridisation, then they are called inner orbital complexes or low spin complexes or spin paired complexes. If the nd orbitals are involved in hybridisation, then such complexes are called outer orbital or high spin or spin free complexes.
- The complexes containing a central metal atom with unpaired electron(s) are paramagnetic. If all the electrons are paired, then the complexes will be diamagnetic.
- Ligands such as CO, CN- , en, and NH3 present in the complexes cause pairing of electrons present in the central metal atom. Such ligands are called strong field ligands.
- Greater the overlapping between the ligand orbitals and the hybridised metal orbital, greater is the bond strength.
3.Define crystal field stabilization energy(CFSE)?Ln5
The crystal field stabilisation energy is defined as the energy difference of electronic configurations in the ligand field (ELF)and the isotropic field/barycentre (Eiso).
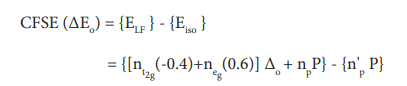
Here,
- nt2g is the number of electrons in t2g orbitals;
- neg is number of electrons in eg orbitals;
- np is number of electron pairs in the ligand field; &
- n’p is the number of electron pairs in the isotropic field (barycentre).
4.Give the difference between double salts and coordination compounds?Ln5
| Double Salt | Co-ordination Compound |
| Double salts contain two simple salt in equimolar proportion | The simple salts from which they are formed may or may not be in equimolar proportion. |
| These exist only in solid state and dissociate into consistent species in their solutions | They retain their identity in solid as well as in solution state. |
| They lose their identity in dissolved state | They do not lose their identity in dissolved state |
| Their properties essentially the same as those of constituent species. | Their properties are different from their constituents for ex: K4[Fe (CN)6] does not of the F e2+ and CN– ions |
| In double salts the metal atom/ion exhibit normal valency | The metal ion exhibits two types of valencies – Primary and secondary |
