p-Block Elements – II
MOST REPEATED QUESTIONS
Number next to question tells how many times the question has been repeated in the past few years
📌 TWO MARKS
1.Write the properties of interhalogen compounds? Any 4 (5)
- The central atom will be the larger one
- It can be formed only between two halogen and not more than two halogens.
- Fluorine can’t act as a central metal atom being the smallest one
- Due to high electronegativity with small size fluorine helps the central atom to attain high coordination number
- They can undergo the auto ionization.
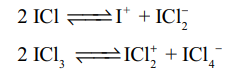
- They are strong oxidizing agents
2.Sulphuric acid is a dibasic acid.Prove it?(4)
It is a strong dibasic acid. as it forms two types of salts namely sulphates and bisulphates.
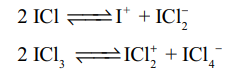
3.Give the uses of Helium?(3)
- It is much less denser than air and hence used for filling air balloons
- Helium and oxygen mixture is used by divers in place of air oxygen mixture. This prevents the painful dangerous condition called bends.
- Helium is used to provide inert atmosphere in electric arc welding of metals
- Helium has lowest boiling point hence used in cryogenics (low temperature science).
📌 THREE MARKS
1.Prove Sulphuric acid is dehydrating agent?(3)
- It is highly soluble in water and has strong affinity towards water and hence it can be used as a dehydrating agent
- When dissolved in water, it forms mono (H2SO4 .H2O) and dihydrates (H2SO4 .2H2O) and the reaction is exothermic.
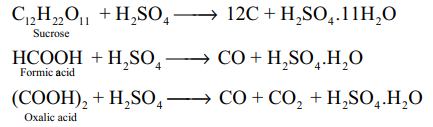
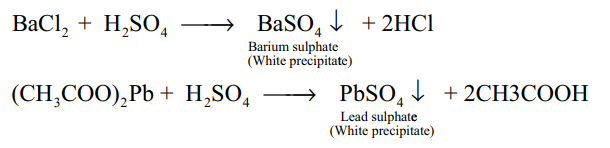
2.Test for sulphuric acid?(2)
Dilute solution of sulphuric acid/aqueous solution of sulphates gives white precipitate (barium sulphate) with barium chloride solution. It can also be detected using lead acetate solution. Here a white precipitate of lead sulphate is obtained.
3.What are interhalogen compounds?Give example(2)
- Each halogen combines with other halogens to form a series of compounds called inter halogen compounds.
- ClF ,ClF3,BrF, BrF3,IF,IF7
4.Explain the action of heat on pcl5?(2)
On heating phosphorous pentachloride, it decomposes into phosphorus trichloride and chlorine.
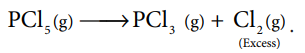
📌 FIVE MARKS
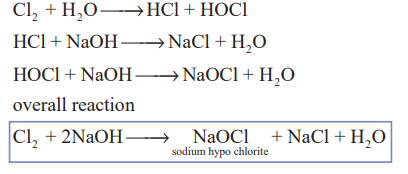
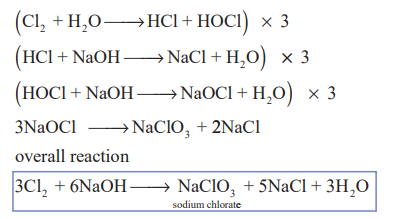
1.Write the action of dil and conc.Naoh on Cl2?(3)
Chlorine reacts with cold dilute alkali to give chloride and hypochlorite while with hot concentrated alkali chlorides and chlorates are formed.
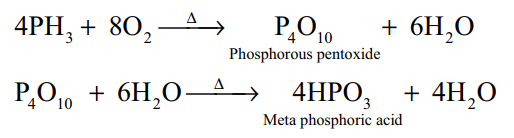
2.Give two equations to illustrate the chemical behaviour of phospine?(2)
Thermal stability: Phosphine decomposes into its elements when heated in absence of air at 317 K or when electric current is passed through it.
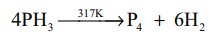
Combustion: When phosphine is heated with air or oxygen it burns to give meta phosphoric acid.
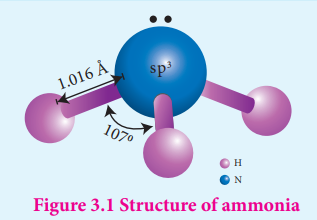
3.Structure of ammonia?(2)
- Ammonia molecule is pyramidal in shape
- N-H bond distance is 1.016 Å and
- H-H bond distance is 1.645 Å with a bond angle 107°.
- The structure of ammonia may be regarded as a tetrahedral with one lone pair of electrons in one tetrahedral position hence it has a pyramidal shape
4.Find the oxidation state of halogen in the following compounds?(2)
1)OF2 (-1 always for flourine)
= -1
2)O2F2
= -1
3)Cl203
- 2x + 3(-2) = 0
- 2x -6 = 0
- 2x = 6
- x = 6/2
- = +3
2)I2O4
- 2x + 4(-2) = 0
- 2x – 8 = 0
- 2x = 8
- X = 8/2
- X = +4
