12th Chemistry Chapter 4 Important 2, 3, 5 Mark Questions 2026
Transition and Inner Transition Elements
MOST REPEATED QUESTIONS
Number next to question tells how many times the question has been repeated in the past few years
📌 TWO MARKS
1.What is lanthanoid contraction?(7)
As we move across from Ce to Lu in 4f series, there is a gradual decrease in atomic and ionic radii of Lanthanoids with increase of atomic number. This decrease in ionic size is called Lanthanoid contraction.
2.Why d block(Transition) elements exhibit variable oxidation state?(4)
- They have more number of electrons in (n-1)d and ns orbitals.
- The energy difference between them (n-1)d and ns orbitals is very small.
3.What are interstitial compounds? (3)
A compound that is formed when small atoms like hydrogen, boron, carbon or nitrogen are trapped in the interstitial hole(empty space) in a metal lattice is called an interstitial compound.
4.Which is more stable? Fe3+or Fe2+– explain.BB13 (3)
Electronic Configuration.
- Fe³+: [Ar] 3d5
- Fe2+: [Ar] 3d6
- Fe³+ ion has half filled d orbital which is more stable than partially filled d orbital of Fe2+.
5.Explain why cr2+ is strongly reducing while MN3+ is strongly oxidizing?(2)
- Cr²+(d4) Readily loses one electron and changes into Cr³+(d³) ion having half-filled t₂glevel.
Thus Cr²+ ion is a strong reducing agent.
- Mn³+(d4) Readily accepts one electron and changed into Mn2+(d5) ion having half-filled d configuration.
Thus Mn3+ ion is a strong oxidising agent.
📌 THREE MARKS
1.Compare lanthanides and actinides || Lanthanoid and Actinoid? (6)
| lanthanides | Actinides |
| Differentiating electron enters in 4f orbital | Differentiating electron eneters in 5f orbital |
| Binding energy of 4f orbitals are higher | Binding energy of 5f orbitals are lower |
| They show less tendency to form complexes | They show greater tendency to form complexes |
| Most of the lanthanoids are colourless | Most of the actinoids are coloured. For example. U3+ (red), U4+ (green) , UO22+ (yellow) |
| They do not form oxo cations | They do form oxo cations such as UO22+ , NpO22+, etc |
| Besides +3 oxidation states lanthanoids show +2 and +4 oxidation states in few cases. | Besides +3 oxidation states actinoids show higher oxidation states such as +4, +5, +6 and +7. |
2.What are the consequences of Lanthanoid contraction?(3)
Consequences of lanthanoid contraction:
- As we move from Ce³+ to Lu³, the basic character of Ln³+ ions decreases. Due to the decrease in the size of Ln³ ions, the ionic character of Ln-OH bond decreases (covalent character 3+ increases) which results in the decrease in the basicity.
- Because of this very small change in radii of lanthanoids, their chemical properties are quite similar.
- The elements of the second and third transition series resemble each other more closely than the elements of the first and second transition series.
3.Write a note on Zeigler – Natta catalyst.Give its uses? (2)
A mixture of TiCl4 and Trialkyl aluminium is called a Zeigler natta catalyst, it is used for polymerization.
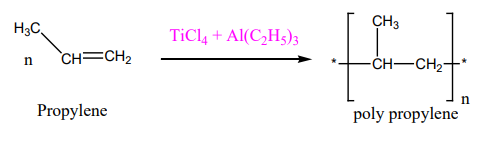
4.Transition metals form complexes or coordination compounds- why?(2)
Transition elements have a tendency to form coordination complexes with ligands. Reasons for complex formation are
- Small size
- High positive charge density.
- Availability of low energy vacant orbitals to accept an electron pairs.
5.Transition metal shows alloy formation.why?(2)
- Transition metals form a number of alloys among themselves, since their atomic sizes are similar and one metal atom can be easily replaced by another metal atom from its crystal lattice to form an alloy.
- The alloys so formed are hard and often have high melting points.
- Examples: Ferrous alloys, gold – copper alloy, chrome alloys etc.,
📌 FIVE MARKS
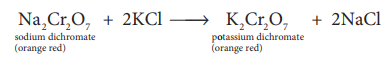
1.How potassium di chromate is prepared from the ore chromite?(2)
Potassium dichromate K2 Cr2 O7
Preparation: Potassium dichromate is prepared from chromate ore. The ore is concentrated by gravity separation. It is then mixed with excess sodium carbonate and lime and roasted in a reverbratory furnace.
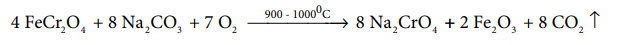
The roasted mass is treated with water to separate soluble sodium chromate from insoluble iron oxide. The yellow solution of sodium chromate is treated with concentrated sulphuric acid which converts sodium chromate into sodium dichromate.
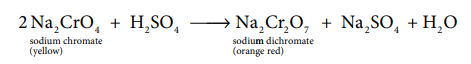
The above solution is concentrated to remove less soluble sodium sulphate. The resulting solution is filtered and further concentrated. It is cooled to get the crystals of Na2 SO4 .2H2 O. The saturated solution of sodium dichromate in water is mixed with KCl and then concentrated to get crystals of NaCl. It is filtered while hot and the filtrate is cooled to obtain K2 Cr2 O7 crystals.